تأثیر خلوص آرگون بر طیفسنجهای خوانش مستقیم طیف کامل: اجازه ندهید ناخالصیها دقت تشخیص را کاهش دهند
Feb 25 , 2026به عنوان یک دستگاه اصلی برای تشخیص عناصر ماده، دقت و پایداری یک طیفسنج خوانش مستقیم طیف کامل، مستقیماً قابلیت اطمینان نتایج آزمایش را تعیین میکند. همه اینها به شدت به "قهرمان نامرئی" در محفظه تحریک - آرگون - وابسته است. به عنوان گاز محافظ و حامل هسته، خلوص آرگون گزینهای نیست که بتواند "زیاد یا کم" باشد، بلکه یک شاخص حیاتی است که بر موفقیت یا شکست آزمایش و طول عمر دستگاه تأثیر میگذارد.
استانداردهای صنعتی، خلوص آرگون ۹۹.۹۹۹٪ (درجه ۵N) را برای این دستگاه الزامی میدانند. اگر خلوص کافی نباشد، ناخالصیهایی مانند اکسیژن، نیتروژن و بخار آب نه تنها منجر به تحریف دادهها میشوند، بلکه به مرور زمان به اجزای اصلی دستگاه نیز آسیب میرسانند. امروز، تأثیرات کلیدی خلوص آرگون را بررسی کرده و راهحلهای عملی ارائه خواهیم داد.

۱. خلوص ناکافی، محیط تحریک را از بین میبرد
طیفسنجهای First برای تحریک نمونهها به تخلیه جرقه/قوس در اتمسفر آرگون متکی هستند. آرگون خالص نمونه را از هوا جدا میکند، اکسیداسیون را مهار میکند و یک کانال تخلیه پایدار را تضمین میکند. نفوذ گازهای ناخالصی مستقیماً این "محیط ایدهآل" را مختل میکند: رایجترین ناخالصیها، اکسیژن و نیتروژن، با اتمهای فلز برانگیخته واکنش میدهند تا اکسیدها و نیتریدها را تشکیل دهند. این امر نه تنها عناصر مورد تجزیه (مانند عناصر به راحتی اکسید شده مانند Si، Al و Mn) را مصرف میکند و منجر به نتایج پایینتر میشود، بلکه باعث قطع یا پرش قوس در نقطه تخلیه نیز میشود که منجر به رنگ جرقه زرد مایل به قرمز و صدای آشفته میشود. از سوی دیگر، بخار آب و دی اکسید کربن، عایق محفظه تحریک را کاهش میدهند و باعث تغییر کانال تخلیه و تداخل بیشتر در فرآیند تحریک میشوند.
دوم. سیگنال و داده: «تداخل مهلک» ناشی از ناخالصیها
برای پرسنل آزمایش، دادههای نادرست و تکرارپذیری ضعیف، بارزترین مشکلات ناشی از ناخالصیهای آرگون هستند:
از یک طرف، گازهای ناخالصی پس از برانگیخته شدن، خطوط طیفی مشخصه خود را تولید میکنند که با خطوط طیفی عناصر مورد تجزیه در نمونه همپوشانی دارند و منجر به تفسیر نادرست دستگاه، سیگنالهای مثبت کاذب یا انحرافات کمی میشوند. از طرف دیگر، ناخالصیها باعث رانش خط پایه و کاهش نسبت سیگنال به نویز میشوند. سیگنالهای ضعیف از عناصر با محتوای کم (مانند P، S و N) توسط پسزمینه محو میشوند، یا غیرقابل تشخیص میشوند یا منجر به نتایج بسیار نوسانی (بیش از استاندارد انحراف معیار نسبی (RSD)) میشوند و ارزش تشخیصی خود را کاملاً از دست میدهند.
III. فرسودگی و استهلاک طولانی مدت: "قاتل نامرئی" اجزای اصلی ابزار دقیق
خلوص ناکافی آرگون نه تنها بر دادهها تأثیر میگذارد، بلکه به طور خاموش به اجزای ابزار دقیق آسیب میرساند و هزینههای نگهداری را افزایش میدهد: در محیطهای با دمای بالا، اکسیژن و بخار آب، الکترودهای تحریک آلیاژ تنگستن/ایریدیوم را اکسید میکنند و باعث فرسایش نوک و پوسته پوسته شدن میشوند. همزمان، مرحله تحریک سرامیکی را دچار خوردگی میکند و لکههای سرسخت و حتی ترک خوردگی ایجاد میکند. گرد و غبار اکسید تولید شده در حین تحریک نیز به سطح توری و آشکارساز CCD میچسبد و راندمان طیفسنجی و حساسیت تشخیص را کاهش میدهد. تعمیر یا تعویض این اجزا بسیار پرهزینه است.
چهارم. راهنمای عملی: چگونه از مسائل مربوط به طهارت اجتناب کنیم؟
بدون رویههای پیچیده، نکات زیر میتوانند به طور مؤثر خطرات مرتبط با خلوص آرگون را کنترل کنند:
۱. منبع گاز مناسب را انتخاب کنید: استفاده از آرگون با خلوص بالای ۹۹.۹۹۹٪ (۵N) از تولیدکنندگان معتبر را در اولویت قرار دهید، به همراه گزارش آزمایش خلوص. از مخلوط کردن یا تعمیر سیلندرها خودداری کنید. تشخیص عناصر کمیاب نیاز به ارتقاء به ۹۹.۹۹۹۵٪ (۵.۵N) دارد.
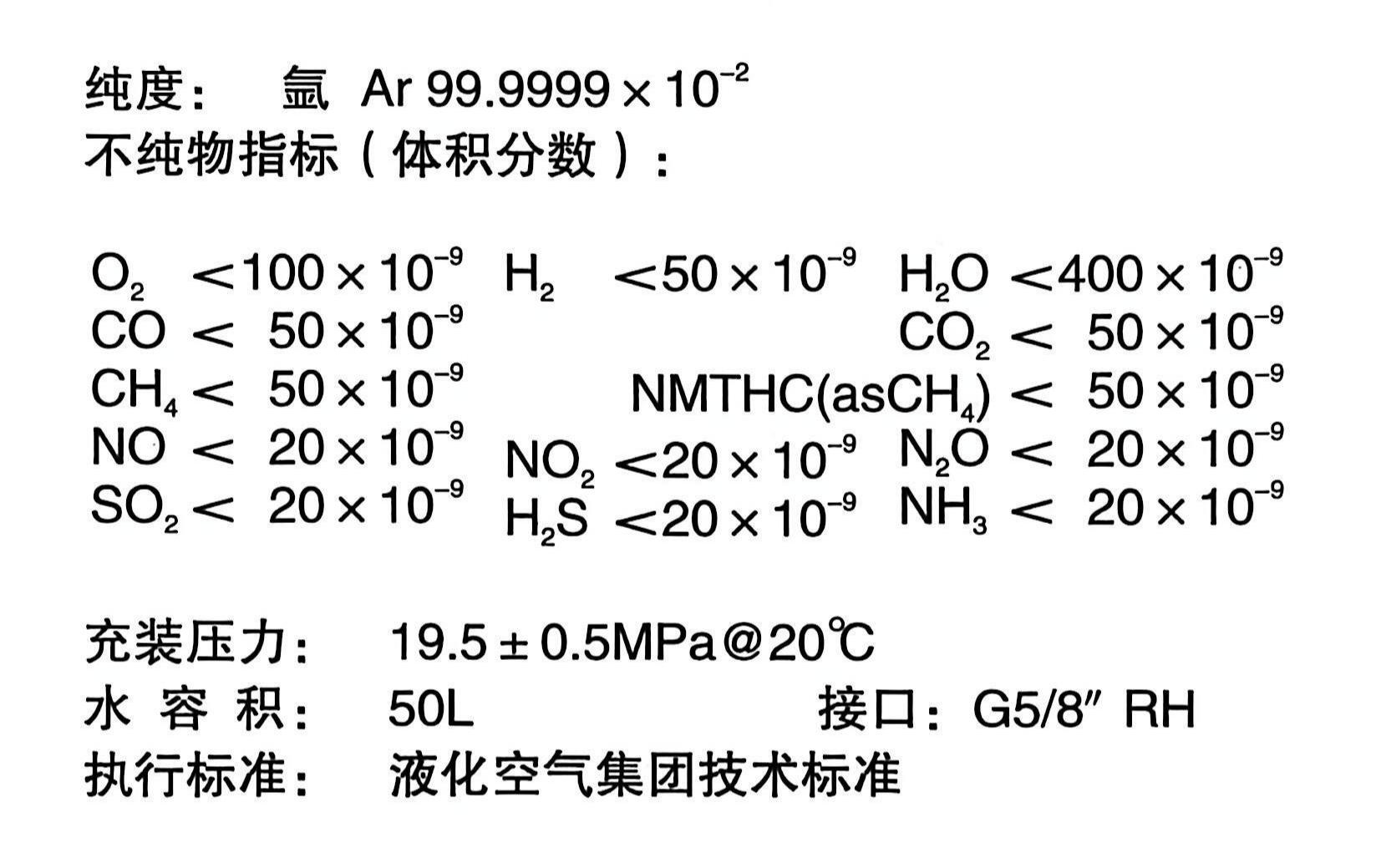
۲. مسیر گاز را بهینه کنید: یک دستگاه تصفیه دو مرحلهای (اکسیژنزدایی + حذف آب + فیلتراسیون) نصب کنید و المنت فیلتر را مرتباً (هر ۳ تا ۶ ماه) تعویض کنید. هنگام تعویض سیلندر گاز جدید، مسیر گاز را به مدت ۱۰ تا ۲۰ ثانیه تمیز کنید تا تمام هوا خارج شود.
۳. استانداردسازی عملیات: قبل از تحریک نمونه، محفظه تحریک را ۵ تا ۱۰ دقیقه قبل از آن تخلیه کنید و فشار باقیمانده در سیلندر گاز را ≥۰.۲ مگاپاسکال حفظ کنید تا از ورود ناخالصیها به مسیر گاز جلوگیری شود.
۴. نگهداری منظم: الکترود تحریک و مرحله تحریک سرامیکی را هر ۱-۲ ماه یکبار تمیز کنید و سالانه نشتی مسیر گاز را بررسی کنید و حلقههای آببندی قدیمی را تعویض کنید.
خلاصه گاز آرگون مانند "خون" یک ... طیفسنج خوانش مستقیم طیف کامل گاز آرگون با خلوص بالای درجه 5N برای تضمین دقت تشخیص و طول عمر دستگاه ضروری است. خلوص ناکافی کوتاهمدت میتواند منجر به تحریف دادهها شود، در حالی که خلوص ناکافی درازمدت میتواند آسیب جبرانناپذیری به قطعات وارد کند. ترکیب کنترل خلوص آرگون، خالصسازی مسیر گاز و نگهداری معمول برای تضمین عملکرد پایدار دستگاه و جلوگیری از ایجاد «مشکلات بزرگ» توسط «ناخالصیهای کوچک» ضروری است.
لطفا برای سوالات بیشتر یا اطلاعات دقیق با ما تماس بگیرید!
تلفن: +86-183-5283 6805
ایمیل: sales@jinyibo.com